Subcellular Structure & Organelle Metabolomics Solution
Online Inquiry- Service Details
- Case Study
Subcellular structures and organelles are the specialized compartments within eukaryotic cells, each with specific functions critical to cellular processes. These structures include mitochondria, nuclei, endoplasmic reticulum, Golgi apparatus, lysosomes, and peroxisomes, among others. Metabolomics provides a unique opportunity to investigate the dynamic metabolic processes occurring within these organelles. Metabolomics at the subcellular level can be used to study the dynamic metabolic processes in specific organelles and avoid the interference caused by metabolite levels and changes in the cytoplasm, and to study and reveal the law of metabolite transfer between organelles and cytoplasm.
Specific Metabolomics Solutions
At Creative Proteomics, our Subcellular Structure & Organelle Metabolomics analysis is a comprehensive suite of services that allows you to explore the metabolic intricacies within various subcellular compartments and organelles. We offer a wide range of analytical capabilities, covering numerous subcellular structures and organelles. Our analysis includes, but is not limited to, the following specific subcellular structures and organelles:
Nucleus Metabolomics: Journey into the heart of the cell and discover the metabolic secrets of the nucleus. Uncover vital metabolites and pathways influencing genetic regulation.
Mitochondria Metabolomics: Explore the powerhouses of the cell. Gain deep insights into energy production and cellular respiration within mitochondria.
Endoplasmic Reticulum (ER) Metabolomics: Decode the ER's role in protein synthesis and lipid metabolism. Understand ER-specific metabolites and their impact.
Golgi Apparatus Metabolomics: Investigate the Golgi apparatus and its involvement in protein modification, secretion, and associated metabolic changes.
Lysosomes Metabolomics: Dive into cellular waste disposal with a focus on lysosomal metabolites. Understand their role in autophagy and degradation processes.
Peroxisomes Metabolomics: Uncover the metabolic nuances of peroxisomes. Learn about their involvement in lipid metabolism and detoxification.
Exosomes Metabolomics: Explore the world of exosome metabolites and their significance in intracellular trafficking and signaling pathways.
Plasma Membrane Metabolomics: Investigate metabolites associated with the cell's outer membrane. Gain insights into membrane lipid composition and signaling processes.
Chloroplast Metabolomics: For plant cells, our services include the analysis of chloroplast metabolic pathways, photosynthesis-related metabolites, comparative network analysis, and evolutionary studies.
Metabolomics Analysis Categories
Metabolite Identification:
Metabolite identification is at the core of metabolomics analysis. It involves characterizing metabolites within specific subcellular structures and organelles. Utilizing mass spectrometry (MS) and nuclear magnetic resonance (NMR) spectroscopy, we unveil the molecular structures of metabolites. This process sheds light on subcellular composition and metabolic activities.
Metabolite Quantification:
Precise quantification reveals the abundance of metabolites within subcellular structures and organelles. This quantitative insight allows us to understand how metabolite levels change with different cellular conditions.
Metabolic Pathway Analysis:
Metabolic pathway analysis delves into the intricate metabolic pathways within subcellular compartments. We decipher how specific metabolites participate in these pathways and elucidate their functions within organelles. This analysis employs advanced bioinformatics tools to map metabolites to specific pathways.
Dynamic Metabolomics:
Cellular responses are dynamic, and so is our approach. Dynamic metabolomics monitors metabolite profiles over time within subcellular structures and organelles. By capturing the kinetics of metabolic processes, we unveil regulatory mechanisms and observe how metabolite levels fluctuate in response to various cellular events.
Metabolite Localization:
To visualize metabolite distribution within organelles, we employ cutting-edge techniques like imaging mass spectrometry (IMS) and subcellular fractionation. These methods unveil the subcellular organization of metabolism and provide insights into organelle-specific metabolite processing.
Functional Metabolomics:
Our approach goes beyond identification and quantification. Functional metabolomics explores how metabolites impact cellular functions. We investigate the physiological roles of metabolites within subcellular structures and organelles, shedding light on the functional significance of metabolic pathways and their influence on cellular responses
Metabolomics Analytical Techniques
Liquid Chromatography-Mass Spectrometry (LC-MS):
- Instrument Model: Thermo Fisher Scientific Q Exactive Plus
Gas Chromatography-Mass Spectrometry (GC-MS):
- Instrument Model: Agilent 7890A GC System coupled with Agilent 5977B MSD
High-Performance Liquid Chromatography-Mass Spectrometry (HPLC-MS):
- Instrument Model: Waters ACQUITY UPLC System coupled with Waters Xevo G2-XS QTOF MS
Triple Quadrupole Mass Spectrometry (QQQ-MS):
- Instrument Model: SCIEX Triple Quad 5500 LC-MS/MS System
Quadrupole Time-of-Flight Mass Spectrometry (QTOF-MS):
- Instrument Model: Agilent 6545 LC/Q-TOF System
Applications of Subcellular Structure & Organelle Metabolomics
Organelle-Specific Metabolite Profiling: Gain insights into the unique metabolic profiles of organelles like mitochondria, the endoplasmic reticulum, and the Golgi apparatus. Identify organelle-specific metabolites and unravel their roles in cellular processes.
Drug Discovery and Development: Accelerate drug discovery by assessing the impact of compounds on organelle-specific metabolism. Identify potential drug targets within specific subcellular compartments, paving the way for targeted therapies.
Cellular Signaling Pathways: Explore the metabolites involved in crucial cellular signaling pathways within organelles. Understand how specific metabolites regulate processes like apoptosis, autophagy, and cell cycle control
Toxicology and Environmental Studies: Investigate the effects of toxins, pollutants, and environmental factors on organelle-specific metabolism. Assess cellular responses to environmental stressors.
Disease Mechanisms: Uncover the metabolic alterations associated with various diseases, including cancer, metabolic disorders, and neurodegenerative conditions. Identify potential biomarkers for disease diagnosis and monitoring.
Functional Genomics: Integrate Subcellular Structure & Organelle Metabolomics with genomics and proteomics data to obtain a holistic view of cellular function. Understand how genetic variations impact organelle-specific metabolism.
Biotechnology and Bioprocessing: Optimize bioproduction processes by analyzing the metabolism of organelles involved in protein synthesis and secretion. Enhance bioprocess efficiency and product yield.
Metabolic Engineering: Engineer cellular metabolism within specific organelles to enhance the production of biofuels, pharmaceuticals, and other valuable compounds.
Agricultural and Plant Science: Study organelle-specific metabolism in plants to improve crop yield, quality, and stress resistance.
Basic Research: Advance our fundamental understanding of cell biology and metabolism by exploring the intricacies of organelle-specific metabolomes.
Bioinformatics Analysis for Metabolomics Service
| Bioinformatics Analysis Aspect | Description |
|---|---|
| Data Preprocessing | Raw data preprocessing, including noise reduction, peak alignment, and data normalization to ensure data quality and comparability. |
| Metabolite Identification | Utilization of databases and spectral matching to identify metabolites based on mass spectrometry data. |
| Metabolite Quantification | Calculation of metabolite concentrations using peak intensities or areas in mass spectrometry data. |
| Pathway Analysis | Enrichment analysis and mapping of identified metabolites to biological pathways to understand their functional relevance. |
| Statistical Analysis | Statistical tests (e.g., t-tests, ANOVA) to identify significant differences in metabolite profiles between samples or groups. |
| Multivariate Analysis | Principal component analysis (PCA), partial least squares-discriminant analysis (PLS-DA), and other multivariate methods to visualize sample clustering and discrimination. |
| Metabolic Network Analysis | Construction and analysis of metabolic networks to identify key metabolic hubs and pathways. |
The subunit structure or analysis item you want to analyze is not within the above range? Please contact us. Our professionals will proactively contact you after receiving your email to understand your needs and provide you with assistance.
Case. Comprehensive Characterization of CD3(+) and CD3(−) Exosomes from Human Plasma
Background
Exosomes, small extracellular vesicles (sEVs) derived from cells, play a crucial role in intercellular communication and are potential candidates for liquid biopsy applications. This study focuses on characterizing CD3(+) exosomes derived from T cells and CD3(−) exosomes from other cell types in human plasma.
Samples
The study involved blood samples from 10 healthy donors (HDs), and plasma was isolated and stored at -80°C. Exosomes were isolated using size-exclusion chromatography (SEC) and separated into CD3(+) and CD3(−) fractions using an immune capture method with anti-CD3 monoclonal antibodies.
Technical Methods
Isolation of Total sEVs from Human Plasma:
- Plasma samples were processed using ultrafiltration and SEC.
- Fraction #4, enriched in intact sEVs, was collected.
- Vesicle size, morphology, and protein content were determined.
Isolation of CD3(+) Exosomes Using Immune Capture:
- Biotin-labeled anti-CD3 mAbs were used to capture CD3(+) exosomes.
- Streptavidin-coated magnetic beads were employed for exosome capture.
- On-bead flow cytometry confirmed CD3 antigen enrichment.
Sample Preparation for Metabolomics and Proteomics Analyses:
- CD3(+)exo and CD3(−)exo samples were prepared.
- Metabolites were analyzed using a targeted quantitative approach.
- Proteomics analysis used LC-MS/MS for protein identification and quantification.
- Protein abundance was estimated based on peptide intensity and normalization to total ion current.
Statistical Analyses:
- Wilcoxon signed-rank tests were used to assess significant differences in protein/metabolite levels.
- The chi-squared independence test examined absence/presence status of compounds.
- FDR correction was applied when necessary.
- Protein interaction networks were analyzed using the STRINGdb database.
Results
Separation of CD3(+) and CD3(−) Vesicles:
- Total plasma sEVs were successfully isolated and characterized as exosomes.
- CD3(+) exosomes were enriched using anti-CD3 mAbs, while CD3(−) exosomes lacked CD3 antigens.
Comparison of the Protein Contents of CD3(+) and CD3(−) Vesicles:
- Proteomic analysis identified 418 proteins, excluding high-abundance plasma proteins.
- Significant differences in protein abundance were observed between CD3(+)exo and CD3(−)exo fractions.
- Immune-related proteins differed in association with specific immune cell types.
- Overrepresented functions/processes were linked to immune responses and stress.
Comparison of the Lipid and Small Metabolites Content of CD3(+) and CD3(−) Vesicles:
- Metabolomic analysis identified 338 metabolites, including lipids and small molecules.
- Differences in metabolite abundance were noted between CD3(+)exo and CD3(−)exo fractions.
- Lipid profiles reflected characteristics of T cell membranes.
Conclusions:
- CD3(+) exosomes can be effectively separated from other plasma sEVs using anti-CD3 antibodies.
- Proteomic and metabolomic analysis revealed distinct molecular features specific to T cells in CD3(+) exosomes.
- CD3(+) exosomes may serve as a valuable "T cell biopsy" for studying T cell-related conditions.
- CD3(−) exosomes provide insights into the activation or functional status of other immune and non-immune cells in circulation.
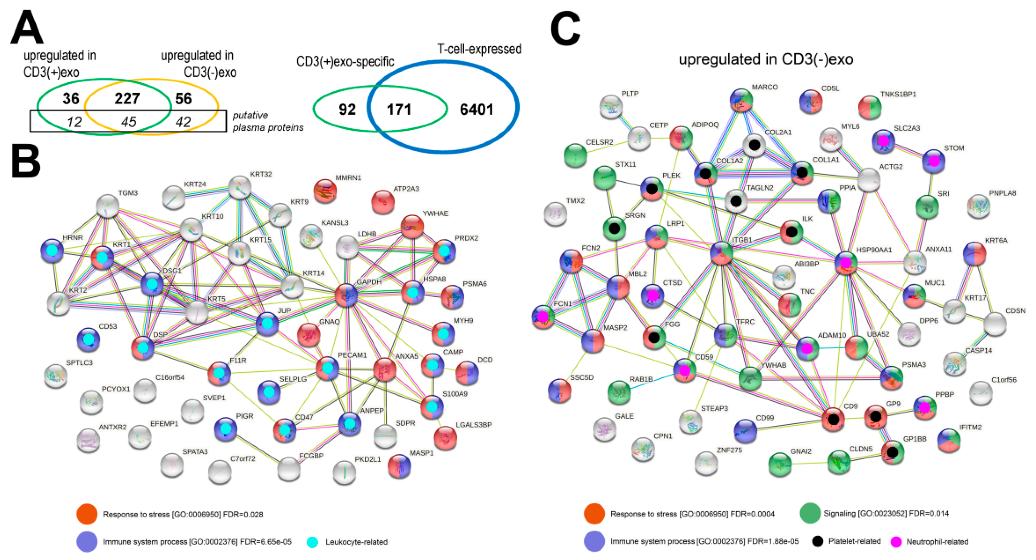 The proteins that were identified in the CD3(+) and CD3(−) fractions of the sEVs from human plasma
The proteins that were identified in the CD3(+) and CD3(−) fractions of the sEVs from human plasma
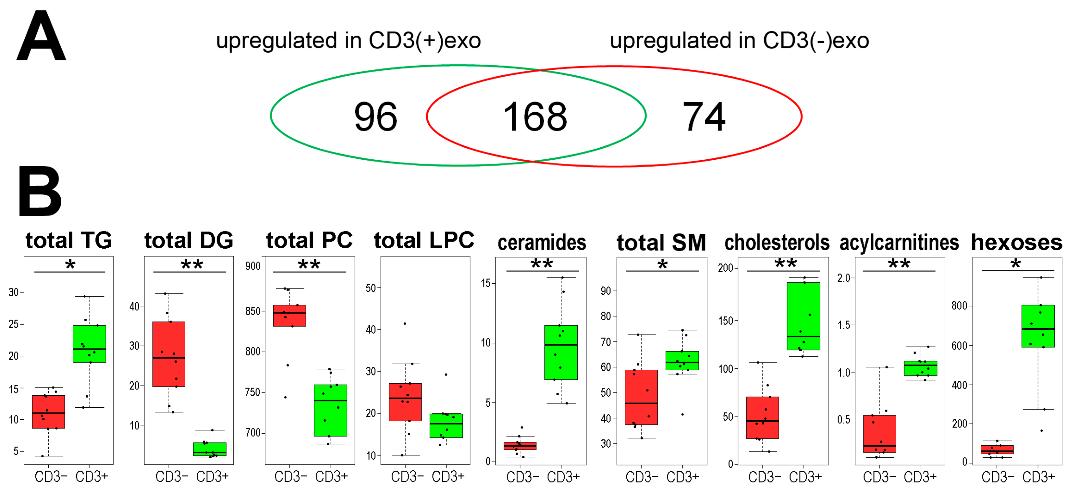 The metabolites that were identified in the CD3(+) and CD3(−) fractions of the sEVs from human plasma
The metabolites that were identified in the CD3(+) and CD3(−) fractions of the sEVs from human plasma
Reference
- Zebrowska, Aneta, et al. "Proteomic and metabolomic profiles of T cell-derived exosomes isolated from human plasma." Cells 11.12 (2022): 1965.
* For Research Use Only. Not for use in diagnostic procedures.