- Service Details
- Case Study
What is pull down?
The pull-down assay is also called binding assay in vitro. It is a commonly used technique for analyzing direct protein-protein interactions (PPIs), which shares similarities with co-immunoprecipitation in terms of methodology. However, instead of employing antibodies, it purifies a tagged target protein act as a "bait" protein to selectively capture its interacting proteins. The pull-down assay is applicable for validating any known PPIs when the proteins of interest can be expressed as a fusion protein with one tag or two tags and isolated with high purity. Furthermore, it serves as a valuable tool for identifying previously unknown binding partners of target proteins, particularly when combined with liquid chromatography tandem mass spectrometry (LC-MS/MS) methods.
The investigation of PPIs is of utmost significance in unraveling the enigma of life. Proteins carry out various functions in life activities, are important part of the cell. Importantly, they do not work alone in the process of life, but constantly interacting with each other proteins, and eventually form a large and complex network of PPIs. Therefore, verifying whether these two proteins interact will give us more insight into the inner workings of cells.
The basic workflow of pull-down assay
This method begins with the expression of a tagged protein (e.g., glutathione S-transferase/GST-, maltose-binding protein/MBP-, biotin-, or His-tagged, etc.) as "bait" protein purified by affinity agarose resin or magnetic beads (e.g., glutathione or cobalt chelate) followed by incubating with "prey" protein derived from various sources, including cell lysates, purified proteins, expression systems, and in vitro transcription/translation systems. After a series of wash buffers to remove unbound proteins, the protein complexes formed between the "bait" and "prey" are subsequently eluted and subjected to SDS-PAGE, western blot (WB), and LC-MS/MS analysis. Most importantly, the inclusion of a parallel control is essential in order to minimize false positive proteins and effectively exclude nonspecific binding protein. There are mainly two methods for the control experiment. First, purify a blank tag such as GST or Biotin immobilized on the affinity agarose resin or magnetic beads to incubate with "prey" proteins. Second, incubate "prey" proteins with blank affinity agarose resin or magnetic beads in parallel.
 Figure 1. Schematic representation of the pull-down procedure.
Figure 1. Schematic representation of the pull-down procedure.
The applications of pull-down assay
1. Detect or confirm the direct interactions between proteins or protein complex.
2. Verify predicted or novel protein-protein interactions.
3. Analyze and quantify the specific isoforms of or active/inactive protein in the tested samples.
Our pull-down assay service includes:
1. Construction of recombinant expression plasmid (Supply the plasmid if obtain).
Table 1. The available fusion tag types for the "bait" protein.
| Tag for "Bait" protein | Affinity ligand |
|---|---|
| Glutathione S-transferase (GST) | Glutathione |
| Poly-histidine (6 × His) | Nickel or cobalt chelate complexes |
| Biotin | Streptavidin |
| Maltose-binding protein (MBP) | Maltose |
| Tandem tags (For instance, His + Biotin tags) to ensure the high purity of the target protein. | |
2. "Bait" protein expression and purification (Purify in a high purity).
3. "Prey" protein extraction.
4. Pull down in parallel with control group and experimental group (After the experiment is optimized, the formal experiment is carried out).
5. Identification of PPIs by SDS-PAGE, WB, and LC-MS/MS technologies. (High quality, precision, and confident).
6. Data processing and report preparation (Fast and accurate).

With over a decade of experimental experience, Creative Proteomics possesses the professional expertise and proficiency to provide a customized experimental scheme that aligns precisely with your specific requirements and projects, ensuring the successful execution of pull-down assays for optimal investigation of PPIs. Our experienced proteomics technical team, strict quality control system, together with ultra-high resolution detection system and professional data pre-processing and analysis capability, ensuring high-quality and reliable results for PPIs proteomics analysis.
Types of pull-down service we provide
Technologies platform
- Professional detection and analysis capability: Experienced research team, strict and skillful techniques.
- High adaptability: The technology is also applicable for the analysis of PTMs and so on.
- High stability and reproducible: Reducing technical variation in the experimental workflow, obtain consistent and reproducible inter- and intra- assay results for data analysis.
- High specificity: Low background and low false positive proteins.
- High resolution and sensitivity: Triple TOF 5600, Q-Exactive, Q-Exactive HF, Orbitrap Fusion™ Tribrid™, etc.
How to place an order
Please feel free to contact us via email for a comprehensive discussion regarding your specific requirements. Our customer service representatives are available round the clock, seven days a week.
Reference
- Chongyi Xu. Pull-down and Co-immunoprecipitation Assays of Interacting Proteins in Plants[J]. Chinese Bulletin of Botany, 2020, 55(1): 62-68.
Lgr5-mediated p53 Repression through PDCD5 leads to doxorubicin resistance in Hepatocellular Carcinoma
Journal: Theranostics
Published: 2019
Main Technology: two-hybrid analysis, Co-immunoprecipitation (Co-IP) and Glutathione-S-transferase (GST) pull-down.
Abstract
The devastating prognosis of hepatocellular carcinoma (HCC) is partially attributed to chemotherapy resistance. Accumulating evidence suggests that the epithelial-mesenchymal transition (EMT) is a key driving force of carcinoma metastasis and chemoresistance in solid tumors. Leucine-rich repeat-containing G protein-coupled receptor 5 (Lgr5), as an EMT inducer, is involved in the potentiation of Wnt signaling in HCC. This study proposes uncovering the roles of Lgr5 in Doxorubicin (Dox) resistance of HCC to improve treatment efficacy for HCC.
Methods: We investigated the expression and significance of Lgr5 in HCC tissue and different cell lines. The effect of Lgr5 in EMT and Dox resistance was analyzed in HCC cells and implanted HCC tumor models. A two-hybrid analysis, using the Lgr5 gene as the bait and a HCC cDNA library, was used to screen targeted proteins that interact with Lgr5. The positive clones were identified by coimmunoprecipitation (Co-IP) and Glutathione-S-transferase (GST) pull-down. The impact of the interaction on Dox resistance was investigated by a series of assays in vitro and in vivo.
Result: We found that Lgr5 was upregulated and positively correlated with poor prognosis in HCC. Additionally, it functioned as a tumor promoter to increase cell migration and induce EMT in HCC cells and increase the resistance to Dox. We identified programmed cell death protein 5 (PDCD5) as a target gene of Lgr5 and we found that PDCD5 was responsible for Lgr5-mediated Dox resistance. Further analysis with Co-IP and GST pull-down assays showed that the N-terminal extracellular domain of Lgr5 could directly bind to PDCD5. Lgr5 induced p53 degradation by blocking the nuclear translocation of PDCD5 and leading to the loss of p53 stabilization. Lgr5 showed a protection against the inhibition of Dox on the growth of tumor subcutaneously injected. Moreover, Lgr5 suppressed Dox-induced apoptosis via the p53 pathway and attenuated the cytotoxicity of Dox to HCC.
Conclusion: Lgr5 induces the EMT and inhibits apoptosis, thus promoting chemoresistance by regulating the PDCD5/p53 signaling axis. Furthermore, Lgr5 may be a potential target gene for overcoming Dox resistance.
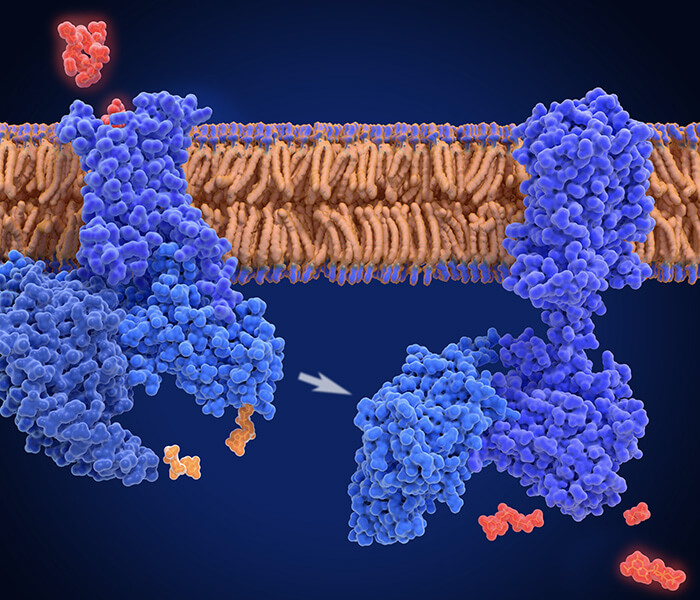
 Figure 1. A graphical model for Lgr5-mediated Dox resistance in HCC cell lines.
Figure 1. A graphical model for Lgr5-mediated Dox resistance in HCC cell lines.