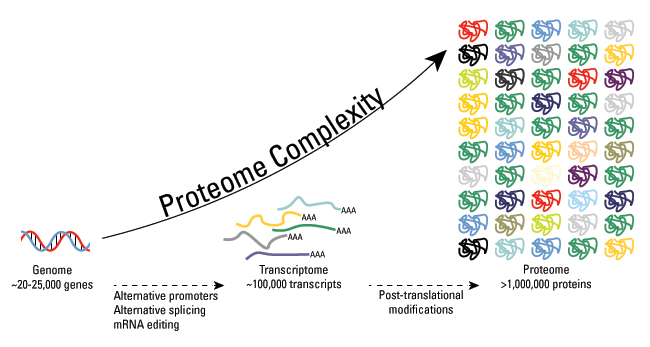
As we know, the human gene set was estimated at about 25,000 genes, while the total number of proteins is expected to be much larger and estimated at over 1 million. The single genes can encode multiple proteins. In addition to alternative splicing mRNA editing as a source of protein complexity, post-translational modifications (PTMs) of proteins can further facilitate the complexity from the level of the genome to the proteome.
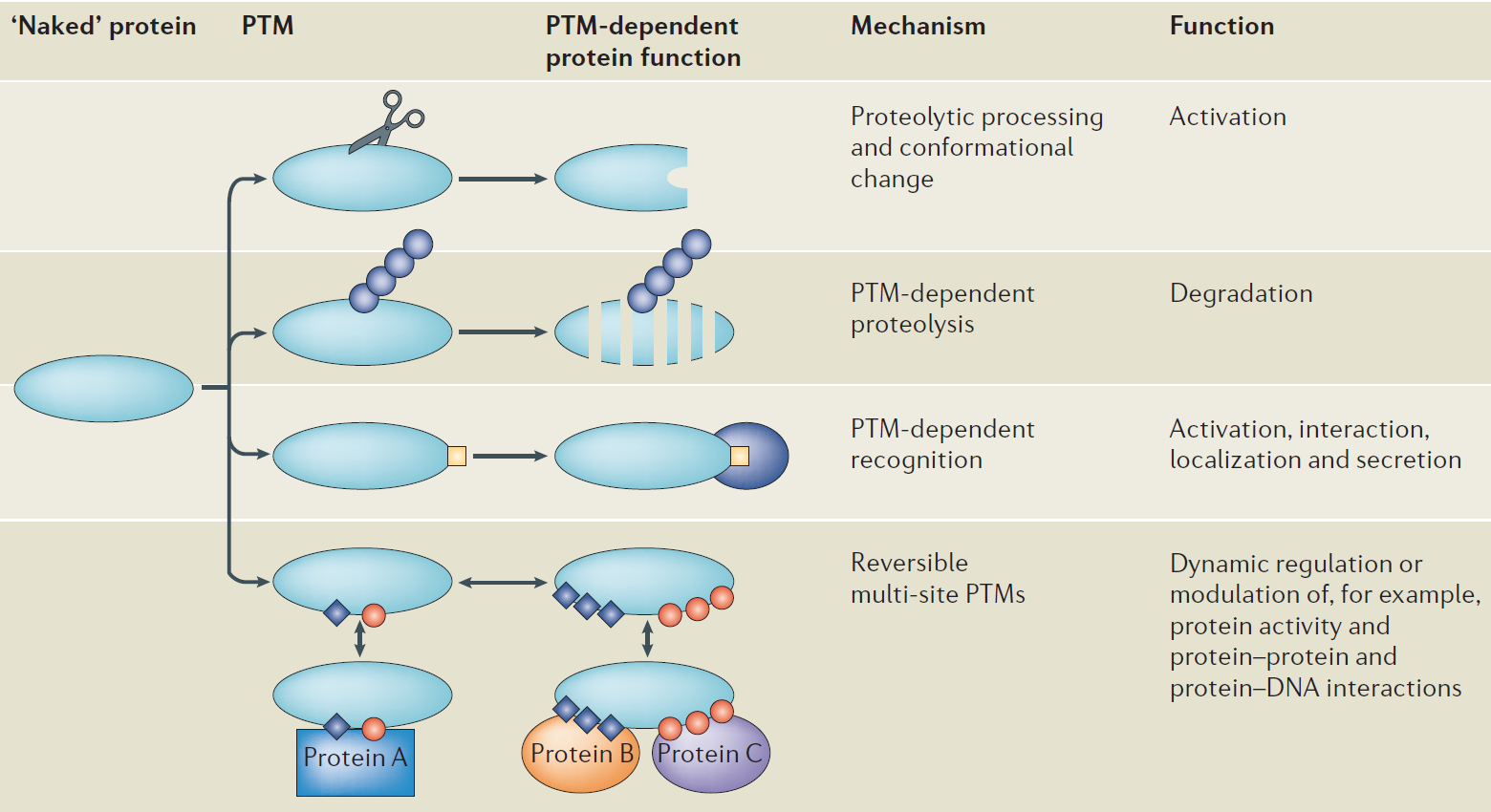
PTMs are chemical alterations to protein structure, typically catalyzed by exceedingly substrate-specific enzymes, which themselves are under strict control by PTMs. Due to a lot of types of PTMs which are covalently attached to amino-acid residues of proteins, there is a rich diversity of gene products. PTMs have a role in different ways. Proteolytic processing and conformational change lead to the activation through cleavage of different sites in the protein. In addition, PTMs can tag proteins for destruction like polyubiquitylation-mediated degradation of proteins. Moreover, PTMs can lead to activation, interaction, localization, and secretion by PTM-dependent recognition. PTMs might induce conformational changes or form a docking site to mediate molecular recognition and stabilize protein–ligand and protein-protein interactions. By reversible multi-site PTMs, they can rapidly and dynamically regulate or modulation protein activities, protein-protein and protein-DNA interactions.
Figure 1. Mechanism of action of post-translational modifications (Jensen O N, 2006)
There are over 200 types of PTMs which been identified. They can affect many aspects of cellular functionalities, like metabolism, signal transduction, and protein stability. Next, we will briefly introduce some common PTMs, including phosphorylation, glycosylation, methylation, acetylation, and ubiquitination.
Phosphorylation
Protein phosphorylation, in which an amino acid residue is phosphorylated by a protein kinase by the addition of a covalently bound phosphate group, is the most widespread type of PTM used in signal transduction. Phosphorylation is done by the kinases and dephosphorylation is done by phosphatases that play opposite roles. In eukaryotic cells, about 86.4% of the protein phosphorylation events occur on serine (Ser or S), while 11.8% protein phosphorylation events occur on threonine whereas only 1.8% on tyrosine residues. Protein phosphorylation plays an extremely important role in many processes, including mediating metabolism, transcription, cell-cycle progression, differentiation, and so on.
Glycosylation
Glycosylation, the attachment of sugar moieties to proteins, is critical for a wide range of biological processes in the cell. There are different types of glycosylation. N-linked glycosylation involves the attachment of the sugar molecule to a nitrogen atom, which is the amide nitrogen of an asparagine (Asn) residue of a protein. O-linked glycosylation is the attachment of a sugar molecule to an oxygen atom in an amino acid residue of the protein. C-mannosylation involves attachment of an α-mannosyl residue to C-2 of the Trp through a C-C bond. Phosphoglycosylation involves the attachment of sugar to a protein through a phosphodiester bond. Glypiation is a special form of glycosylation, in which a protein is attached to a lipid anchor.
Methylation
Protein methylation involves the addition of a methyl group to a protein amino acid. Arginine methylation can work as an epigenetic regulator of transcription and play important roles in pre-mRNA splicing, DNA damage signaling, mRNA translation, and cell signaling. It is reported that methylation can occur on the side chains of at least nine of the twenty common amino acid residues, including arginine, lysine, histidine, methionine, cysteine, glutamine, asparagine, glutamic acid, and aspartic acid. Among them, arginine and lysine are by far the most commonly methylated residues. In addition, methylation of arginine or lysine does not affect the overall charge of these residues, and their side chains remain positively charged even when methylated. Lysine residue can be methylated up to three times and an arginine residue up to two times. The methyl group is one of the smallest PTMs and affects relatively little to the ‘steric bulk’ of the modified side chains.
Acetylation
Protein acetylation involves the process that the acetyl group from acetyl coenzyme A (Ac-CoA) is transferred to a specific site on a polypeptide chain. Proteins can be acetylated by both enzymatic and non-enzymatic processes. In humans, 80–90% of all proteins become co-translationally acetylated at their N-termini of the nascent polypeptide chains. An acetyl group can be transferred from Ac-CoA to the N-terminal amino group of a polypeptide, catalyzed by N-terminal acetyltransferases (NATs). Reversible acetylation of the ε-amino group of a lysine residue is the other common type, which is catalyzed by lysine acetyltransferases (KATs) and lysine deacetylases (KDACs). In addition, acetylation interferes with metabolic processes and energy homeostasis owing to the consumption of Ac-CoA during acetylation and NAD+ during deacetylation by specific KDACs.
Ubiquitination
Ubiquitination involves the attachment of the polypeptide ubiquitin to target proteins by a set of three enzymes, including ubiquitin-activating enzyme E1, ubiquitin-conjugating enzyme E2, and ubiquitin ligase E3. The E1 enzyme first activates the carboxy terminus of the ubiquitin molecule, using the energy from converting an ATP molecule to AMP and pyrophosphate (PPi). The activated ubiquitin is attached to the sulfur of the E1 active-site cysteine residue. Ubiquitin is then transferred from E1 to E2, and E3 facilitates the transfer of ubiquitin from E2 to the substrate protein. Ubiquitination can mark proteins for degradation via the proteasome, alter their cellular location, affect their activity, and promote or prevent protein interactions.
Analysis of protein PTMs, including the modification types and modified sites, is particularly important for the study of cell biology as well as disease diagnostics and prevention.
References:
- Jensen O N. Interpreting the protein language using proteomics. Nature reviews Molecular cell biology, 2006, 7(6): 391.
- Spiro R G. Protein glycosylation: nature, distribution, enzymatic formation, and disease implications of glycopeptide bonds. Glycobiology, 2002, 12(4): 43R-56R.
- Murn J, Shi Y. The winding path of protein methylation research: milestones and new frontiers. Nature Reviews Molecular Cell Biology, 2017, 18(8): 517.
- Wang Y C, Peterson S E, Loring J F. Protein post-translational modifications and regulation of pluripotency in human stem cells. Cell research, 2014, 24(2): 143.
- Drazic A, Myklebust L M, Ree R, et al. The world of protein acetylation. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics, 2016, 1864(10): 1372-1401.